There’s considerable excitement that 3D printing technology might one day allow scientists to produce fully functional replacement organs from one’s own cells. While there’s still a lot to learn, this video shows just some of the amazing progress that’s now being made.
The video comes from a bioengineering team at Rice University, Houston, that has learned to bioprint the small air sacs in the lungs. When hooked up to a machine that pulsed air in and out of the air sacs, the rhythmic movement helped to mix red blood cells traveling through an associated blood vessel network. Those red cells also took up oxygen in much the way that blood vessels do when surrounding the hundreds of millions of air sacs in our lungs.
As mentioned in the video, one of the biggest technical hurdles in growing fully functional replacement tissues and organs is to find a way to feed the growing tissues with a blood supply and to remove waste products. In this study recently published in Science [1], the NIH-supported team cleared this hurdle by creating an open-source bioprinting technology they call SLATE, which is short for “stereo-lithography apparatus for tissue engineering.”
The SLATE system “grows” soft hydrogel scaffolds one layer at a time. Each layer is printed using a liquid pre-hydrogel solution that solidifies when exposed to blue light. By also projecting light into the hydrogel as a pixelated 3D shape, it’s possible to print complex 3D structures within minutes.
When the researchers first started, their printouts lacked the high resolution, submillimeter-scale channels needed to generate intricate vascular networks. In other manufacturing arenas, light-absorbing chemicals have helped control the conversion from liquid to solid in a very fine polymer layer. But these industrial light-absorbing chemicals are highly toxic and therefore unsuitable for scaffolds that grow living tissues and organs.
The researchers, including Bagrat Grigoryan, Jordan Miller, and Kelly Stevens, wondered whether they could swap out those noxious ingredients with synthetic and natural food dyes widely used in the food industry. These dyes include curcumin, anthocyanin, and tartrazine (yellow dye #5). Their studies showed that those fully biocompatible dyes worked as effective light absorbers, allowing the scientists to recreate the complex architectures of human vasculature. Importantly, the living cells survived within the soft scaffold!
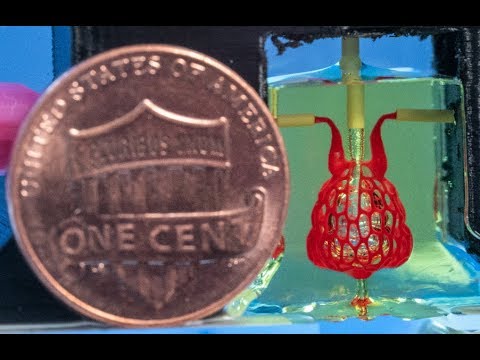
These models are already yielding intriguing new insights into the vascular structures found within our organs and how those architectures may influence function in ways that hadn’t been well understood. In the near term, tissues and organs grown on such scaffolds might also find use as sophisticated, 3D tissue “chips,” with potential for use in studies to predict whether drugs will be safe in humans.
In the long term, this technology may allow production of replacement organs from those needing them. More than 100,000 men, women, and children are on the national transplant waiting list in the United States alone and 20 people die each day waiting for a transplant [2]. Ultimately, with the aid of bioprinting advances like this one, perhaps one day we’ll have a ready supply of perfectly matched and fully functional organs.

